
An old fertilizer spreader

A large, modern fertilizer spreader
Fertilizers are soil amendments applied to promote plant growth; the main nutrients present in fertilizer are nitrogen, phosphorus, and potassium (the 'macronutrients') and other nutrients ('micronutrients') are added in smaller amounts. Fertilizers are usually directly applied to soil, and also sprayed on leaves ('foliar feeding').
Fertilizers are roughly broken up between organic and inorganic fertilizer, with the main difference between the two being sourcing, and not necessarily differences in nutrient content.
Organic fertilizers and some mined inorganic fertilizers have been used for many centuries, whereas chemically-synthesized inorganic fertilizers were only widely developed during the industrial revolution. Increased understanding and use of fertilizers were important parts of the pre-industrial British Agricultural Revolution and the industrial green revolution of the 20th century.

Tennessee Valley Authority: "Results of Fertilizer" demonstration 1942
Fertilizers typically provide, in varying proportions:
- the three primary macronutrients: nitrogen, phosphorus, and potassium.
- the three secondary macronutrients: calcium(Ca), sulfur (S), magnesium (Mg).
- and the micronutrients or trace minerals: boron (B), chlorine (Cl), manganese (Mn), iron (Fe), zinc (Zn), copper (Cu), molybdenum (Mo) and selenium (Se).
The macronutrients are consumed in larger quantities and are present in plant tissue in quantities from 0.2% to 4.0% (on a dry matter weight basis). Micronutrients are consumed in smaller quantities and are present in plant tissue in quantities measured in parts per million (ppm), ranging from 5 to 200 ppm, or less than 0.02% dry weight.[1]
Labeling of fertilizers
Macronutrient fertilizers
Macronutrient fertilizers are labeled with an NPK analysis and also "N-P-K-S" in Australia[2].
An example of labeling for the fertilizer potash is composed of 1:1 potassium to carbonate or 47% potassium and 53% Carbonate by weight (owing to differences in molecular weight between the potassium and carbonate). Traditional analysis of 100g of KCl would yield 60g of K2O. The percentage yield of K2O from the original 100g of fertilizer is the number shown on the label. A potash fertilizer would thus be labeled 0-0-60, not 0-0-52.
History
The modern understanding of plant nutrition dates to the 19th century and the work of Justus von Liebig, among others. Management of soil fertility, however, has been the pre-occupation of farmers for thousands of years.
Type of Fertilizer
Fertilizers come in various shapes and forms. The most typical form is granular fertilizer (powder form), usually come in a bag / box. The next most common form is liquid fertilizer; some advantages of liquid lawn fertilizer are its immediate effect and wide coverage. Moreover, there is also a form of slow-release fertilizer which solves the problem of "burning" the plants due to excessive nutrients. This kind of fertilizer come in various form like fertilizer spikes, tabs, etc. Finally, organic fertilizer is on the rise as people are resorting to a green / environmental friendly products. Although organic fertilizer usually contain less nutrients, some people still prefer organic due to natural ingredients.
Inorganic fertilizer (synthetic fertilizer)
Fertilizers are broadly divided into organic fertilizers (composed of enriched organic matter—plant or animal), or inorganic fertilizers (composed of synthetic chemicals and/or minerals).
Inorganic fertilizer is often synthesized using the Haber-Bosch process, which produces ammonia as the end product. This ammonia is used as a feedstock for other nitrogen fertilizers, such as anhydrous ammonium nitrate and urea. These concentrated products may be diluted with water to form a concentrated liquid fertilizer (e.g. UAN). Ammonia can be combined with rock phosphate and potassium fertilizer in the Odda Process to produce compound fertilizer.
The use of synthetic nitrogen fertilizers has increased steadily in the last 50 years, rising almost 20-fold to the current rate of 1 billion tonnes of nitrogen per year.[3] The use of phosphate fertilizers has also increased from 9 million tonnes per year in 1960 to 40 million tonnes per year in 2000. A maize crop yielding 6-9 tonnes of grain per hectare requires 30–50 kg of phosphate fertilizer to be applied, soybean requires 20–25 kg per hectare.[4] Yara International is the world's largest producer of nitrogen based fertilizers.[5]
Application
Synthetic fertilizers are commonly used to treat fields used for growing maize, followed by barley, sorghum, rapeseed, soy and sunflower[citation needed]. One study has shown that application of nitrogen fertilizer on off-season cover crops can increase the biomass (and subsequent green manure value) of these crops, while having a beneficial effect on soil nitrogen levels for the main crop planted during the summer season.[
Problems of inorganic fertilizer
Trace mineral depletion
Many inorganic fertilizers do not replace trace mineral elements in the soil which become gradually depleted by crops. This depletion has been linked to studies which have shown a marked fall (up to 75%) in the quantities of such minerals present in fruit and vegetables.[8]
However, a recent review of 55 scientific studies concluded "there is no evidence of a difference in nutrient quality between organically and conventionally produced foodstuffs" [9] Conversely, a major long-term study funded by the European Union[10][11][12] found that organically-produced milk and produce were significantly higher in antioxidants (such as carotenoids and alpha-linoleic acids) than their conventionally grown counterparts.
In Western Australia deficiencies of zinc, copper, manganese, iron and molybdenum were identified as limiting the growth of broad-acre crops and pastures in the 1940s and 1950s[citation needed]. Soils in Western Australia are very old, highly weathered and deficient in many of the major nutrients and trace elements[citation needed]. Since this time these trace elements are routinely added to inorganic fertilizers used in agriculture in this state[citation needed].
Overfertilization
Over-fertilization of a vital nutrient can be as detrimental as underfertilization.[13] "Fertilizer burn" can occur when too much fertilizer is applied, resulting in a drying out of the roots and damage or even death of the plant.[14]
[edit] High energy consumption
The production of synthetic ammonia currently consumes about 5% of global natural gas consumption, which is somewhat under 2% of world energy production.[15]
Natural gas is overwhelmingly used for the production of ammonia, but other energy sources, together with a hydrogen source, can be used for the production of nitrogen compounds suitable for fertilizers. The cost of natural gas makes up about 90% of the cost of producing ammonia.[16] The increase in price of natural gases over the past decade, along with other factors such as increasing demand, have contributed to an increase in fertilizer price[17].
Long-Term Sustainability
Inorganic fertilizers are now produced in ways which cannot be continued indefinitely[citation needed]. Potassium and phosphorus come from mines (or saline lakes such as the Dead Sea) and such resources are limited. Atmospheric (unfixed) nitrogen is effectively unlimited (forming over 70% of the atmospheric gases), but this is not in a form useful to plants. To make nitrogen accessible to plants requires nitrogen fixation (conversion of atmospheric nitrogen to a plant-accessible form).
Artificial nitrogen fertilizers are typically synthesized using fossil fuels such as natural gas and coal, which are limited resources. In lieu of converting natural gas to syngas for use in the Haber process, it is also possible to convert renewable biomass to syngas (or wood gas) to supply the necessary energy for the process, though the amount of land and resources (ironically often including fertilzer) necessary for such a project may be prohibitive (see Energy conservation in the United States).
Organic fertilizer

Compost bin for small-scale production of organic fertilizer

A large commercial compost operation
Organic fertilizers include naturally-occurring organic materials, (e.g. manure, worm castings, compost, seaweed), or naturally occurring mineral deposits (e.g. saltpeter, guano).
Benefits of organic fertilizer
In addition to increasing yield[citation needed] and fertilizing plants directly, organic fertilizers can improve the biodiversity (soil life) and long-term productivity of soil[18][19], and may prove a large depository for excess carbon dioxide[20][21][22].
Organic nutrients increase the abundance of soil organisms by providing organic matter and micronutrients for organisms such as fungal mycorrhiza[23], (which aid plants in absorbing nutrients), and can drastically reduce external inputs of pesticides, energy and fertilizer, at the cost of decreased yield[24].
Comparison with inorganic fertilizer
Organic fertilizer nutrient content, solubility, and nutrient release rates are typically all lower than inorganic fertilizers[25][26]. One study[which?] found that over a 140-day period, after 7 leachings:
- Organic fertilizers had released between 25% and 60% of their nitrogen content
- Controlled release fertilizers (CRFs) had a relatively constant rate of release
- Soluble fertilizer released most of its nitrogen content at the first leaching
In general, the nutrients in organic fertilizer are both more dilute and also much less readily available to plants. According to UC IPM, all organic fertilizers are classified as 'slow-release' fertilizers, and therefore cannot cause nitrogen burn[27].
Organic fertilizers from composts and other sources can be quite variable from one batch to the next{http://www.msuorganicfarm.com/Compost.pdf}, without batch testing amounts of applied nutrient cannot be precisely known. Nevertheless they are at least as effective as chemical fertilizers over longer periods of use{http://md1.csa.com/partners/viewrecord.php?requester=gs&collection=TRD&recid=0002290EN&q=http%3A%2F%2Fwww.csa.com%2Fpartners%2Fviewrecord.php%3Frequester%3Dgs%26collection%3DTRD%26recid%3D0002290EN&uid=789131166&setcookie=yes}.
Organic fertilizer sources
Animal

Decomposing animal manure, an organic fertilizer source
Animal-sourced urea , are suitable for application organic agriculture, while pure synthetic forms of urea are not[28][29]. The common thread that can be seen through these examples is that organic agriculture attempts to define itself through minimal processing (in contrast to the man-made Haber process), as well as being naturally-occurring or via natural biological processes such as composting.
Sewage sludge use in organic agricultural operations in the U.S. has been extremely limited and rare due to USDA prohibition of the practice (due to toxic metal accumulation, among other factors)[30][31][32]. The USDA now requires 3rd-party certification of high-nitrogen liquid organic fertilizers sold in the U.S.[33]
Plant
Cover crops are also grown to enrich soil as a green manure through nitrogen fixation from the atmosphere[34]; as well as phosphorus (through nutrient mobilization)[35] content of soils.
Mineral
Naturally mined powdered limestone[36], mined rock phosphate and sodium nitrate, are inorganic (in a chemical sense), are energetically-intensive to harvest, yet are approved for usage in organic agriculture in minimal amounts[36][37][38].
Environmental effects of fertilizer use
Water
Eutrophication
The nitrogen-rich compounds found in fertilizer run-off is the primary cause of a serious depletion of oxygen in many parts of the ocean, especially in coastal zones; the resulting lack of dissolved oxygen is greatly reducing the ability of these areas to sustain oceanic fauna.[39] Visually, water may become cloudy and discolored (green, yellow, brown, or red).
About half of all the lakes in the United States are now eutrophic, while the number of oceanic dead zones near inhabited coastlines are increasing.[40] As of 2006, the application of nitrogen fertilizer is being increasingly controlled in Britain and the United States[citation needed]. If eutrophication can be reversed, it may take decades[citation needed] before the accumulated nitrates in groundwater can be broken down by natural processes.
High application rates of inorganic nitrogen fertilizers in order to maximize crop yields, combined with the high solubilities of these fertilizers leads to increased runoff into surface water as well as leaching into groundwater.[41][42][43] The use of ammonium nitrate in inorganic fertilizers is particularly damaging, as plants absorb ammonium ions preferentially over nitrate ions, while excess nitrate ions which are not absorbed dissolve (by rain or irrigation) into runoff or groundwater.[44]
Blue Baby Syndrome
Nitrate levels above 10 mg/L (10 ppm) in groundwater can cause 'blue baby syndrome' (acquired methemoglobinemia), leading to hypoxia (which can lead to coma and death if not treated)[45].
Soil
Soil acidification
Nitrogen-containing inorganic and organic fertilizers can cause soil acidification when added [46]. [4]. This may lead to decreases in nutrient availability which may be offset by liming.
[edit] Persistent organic pollutants
Toxic persistent organic pollutants ("POPs"), such as Dioxins, polychlorinated dibenzo-p-dioxins (PCDDs), and polychlorinated dibenzofurans (PCDFs) have been detected in agricultural fertilizers and soil amendments[47]
Heavy metal accumulation
The concentration of up to 100 mg/kg of cadmium in phosphate minerals (for example, minerals from Nauru[48] and the Christmas islands[49]) increases the contamination of soil with cadmium, for example in New Zealand.[50]
Uranium is another example of a contaminant often found in phosphate fertilizers (at levels from 7 to 100 pCi/g)[51]. Eventually these heavy metals can build up to unacceptable levels and build up in vegetable produce.[50] (See cadmium poisoning) Average annual intake of uranium by adults is estimated to be about 0.5 mg (500 μg) from ingestion of food and water and 0.6 μg from breathing air[52].
Steel industry wastes, recycled into fertilizers for their high levels of zinc (essential to plant growth), wastes can include the following toxic metals: leadarsenic, cadmium, chromium, and nickel. The most common toxic elements in this type of fertilizer are mercury, lead, and arsenic.[54][55] Concerns have been raised concerning fish meal mercury content by at least one source in Spain[56]
Also, highly-radioactive Polonium-210 contained in phosphate fertilizers is absorbed by the roots of plants and stored in its tissues; tobacco derived from plants fertilized by rock phosphates contains Polonium-210 which emits alpha radiation estimated to cause about 11,700 lung cancer deaths each year worldwide.[57][58] [59][60][61][62]
For these reasons, it is recommended that nutrient budgeting, through careful observation and monitoring of crops, take place to mitigate the effects of excess fertilizer application.
Other problems
Atmospheric effects
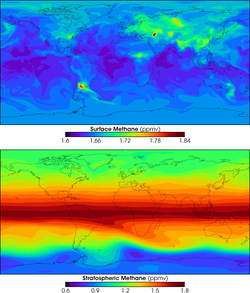
Global methane concentrations (surface and atmospheric) for 2005; note distinct plumes
Methane emissions from crop fields (notably rice paddy fields) are increased by the application of ammonium-based fertilizers; these emissions contribute greatly to global climate change as methane is a potent greenhouse gas.[63]
Through the increasing use of nitrogen fertilizer, which is added at a rate of 1 billion tons per year presently[64] to the already existing amount of reactive nitrogen, nitrous oxide (N2O) has become the third most important greenhouse gas after carbon dioxide and methane. It has a global warming potential 296 times larger than an equal mass of carbon dioxide and it also contributes to stratospheric ozone depletion.[65]
Storage and application of some nitrogen fertilizers in some[which?] weather or soil conditions can cause emissions of the potent greenhouse gas—nitrous oxide. Ammonia gas (NH3) may be emitted following application of 'inorganic' fertilizers and/or manures and slurries.[citation needed]
The use of fertilizers on a global scale emits significant quantities of greenhouse gas into the atmosphere. Emissions come about through the use of:[66]
By changing processes and procedures, it is possible to mitigate some, but not all, of these effects on anthropogenic climate change.[citation needed]
Increased pest health
Excessive nitrogen fertilizer applications can also lead to pest problems by increasing the birth rate, longevity and overall fitness of certain agricultural pests.











